Boron-doped olympicenes are surprisingly stable
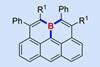
By possessing useful electronic properties, boraolympicenes could have potential applications in organic electronics
Researchers in China have synthesised planar boron-doped olympicenes via a simple one-pot reaction. These boraolympicene compounds exhibit surprising chemical stability and electronic properties that could see them find use within organic electronics.