A highly-efficient cobalt terpyridine-based catalyst has been developed that can convert carbon dioxide into methane. The catalyst operates at the gas-liquid-solid interface improving efficiency, offering a way to turn a waste product into a fuel.
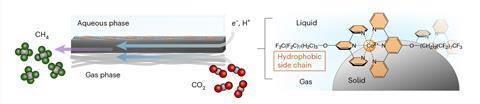
Researchers have long sought efficient ways to convert carbon dioxide into methane to combat the climate crisis. Yet, creating the right chemical conditions has proven tricky due to the different physical states of carbon dioxide and water, as well as water’s tendency to undergo competing side reactions.
Now, research from a team at the Universities of Bonn and Montreal has brought together carbon dioxide and water on a gas diffusion electrode to form methane. Instead of using energy or high pressure to begin the reaction, the researchers use electricity as the driving force. The catalyst first deposited onto the electrode, where it can sequester carbon dioxide in its ‘active centre’, weakening the bonds between the carbon and oxygen atoms. To replace oxygen with hydrogen, while preventing unwanted side reactions, the catalyst incorporates long, hydrophobic perfluorinated alkyl side chains that repel water. These act as a proton conveyor belt, capturing hydrogen atoms from water molecules, shuttling them to the active sites and hydrogenating the intermediate en route to generate methane.
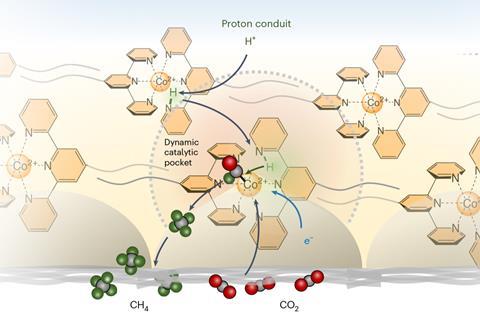
While the process has achieved over 80% efficiency with minimal undesirable side products, the catalyst is not yet suitable for large-scale methane production, owing to the challenge of preventing the electrode from being flooded. Additionally, the reaction efficiency can fluctuate due to changes in repeated drying and hydration of the gas-liquid-solid interface over time. The reaction principles could, however, be adapted to produce other chemicals, such as ethylene, a key precursor in plastics manufacturing, potentially leading to more sustainable plastic production.
References
M McKee et al, Nat. Chem., 2024, DOI: 10.1038/s41557-024-01650-6














2 readers' comments