UK researchers have managed to achieve a particularly tricky substitution on a benzene ring
UK Researchers have discovered that, simply by using a copper catalyst, they are able to perform tricky substitution reactions at a difficult position on benzene rings. The team from the University of Cambridge hope that their method will simplify the synthesis of aromatic molecules both in academia and the pharmaceutical industry.
Aromatic compounds are found throughout organic chemistry, appearing in pharmaceuticals and functional materials as well as in the synthesis of natural products and complex molecules. However, while there are several methods for adding substituents to the ortho- and para- positions of aromatic rings, meta-substitution has always been much trickier.
Matthew Gaunt and his team have found a way to solve this problem by using a simple copper catalyst. The copper salt they used, copper triflate, is cheap, air stable and can be used in mild conditions.
Traditionally, meta-substituted compounds are either generated using aromatics with electron-withdrawing substituents, which reduce the reactivity of the system, or through multi-step syntheses. In contrast, Gaunt’s method allows direct access to these molecules in one simple step. The team found that by reacting amide-substituted benzene rings with phenyl electrophiles in the presence of copper triflate, they were able to generate the meta product exclusively.
’The combination of the copper catalyst with the amide group somehow tricks this molecule into behaving differently to how it wants to,’ says Gaunt. ’If you have an aromatic molecule which is electron rich it tends to react at the ortho or para positions. So it takes the traditional rules of electrophilic aromatic substitution and bends them to get opposite products out to those you would expect.’
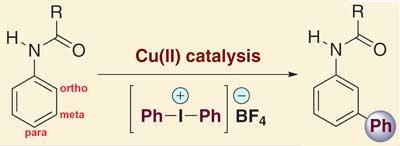
The researchers believe the reaction proceeds via Cu(III), a highly electrophilic species that attacks the electron rich ring, disrupting its aromatic stability and inducing a positive charge. ’The oxygen of the amide group is then attacking that positive charge,’ says Gaunt. ’In doing that, the only way you can involve the oxygen is to put the copper into the meta position,n which is ultimately where the functionalisation happens.’ The aromatic stability of the ring is quickly restored, followed by a reductive elimination step which releases the meta-bound copper and generates the product.
Although the amide plays a crucial part in the reaction mechanism, Gaunt is already investigating whether the reaction will work with other substituents and other electrophiles. ’We want to be able to change the type of reaction - to put fluorine in the meta position or a methyl group or a bromine or an amine - so that we can essentially make any bond that we want.’
The team hopes their research will have a big impact on organic synthesis in the pharmaceutical industry, and for the general synthesis of natural products and complex molecules. ’And because aromatics are so important in pretty much most of everyday life functionalised materials, polymers could certainly make use of the monomeric building blocks which might arise from these reactions,’ adds Gaunt.
Jonathan Clayden, professor of organic chemistry at the University of Manchester, UK, says the team’s method is an important addition to the chemistry toolkit. ’There are a lot of possibilities for where this could go,’ he says. ’It’s building block type chemistry so it will change the way that people think about making aromatic systems.’
’I think the most exciting thing is the potential it offers,’ says Gaunt. ’I hope this will open the door to be able to control aromatic substitution chemistry which maybe breaks the old-fashioned rules.’
Manisha Lalloo
References
Science, vol 323, 2009. DOI: 10.1126/science.1169975






No comments yet