Japanese researchers have created a gel that harnesses free radicals to heal without any outside help
Japanese chemists have made a covalently cross-linked gel that can repair itself simply by bringing the broken surface together. The new material can heal itself even after damaged surfaces have been kept apart for as long as five days.
Inspired by our bodies’ ability to heal, self-healing materials have the ability to repair themselves when they are damaged. That’s important, for example, in medical devices. Smart materials that do this have traditionally been made in two ways, either using supramolecular chemistry like host-guest interactions, which are fast acting but sensitive to water, or covalent bonds that are tolerant to water and many functional groups, but often require energy to reform bonds.
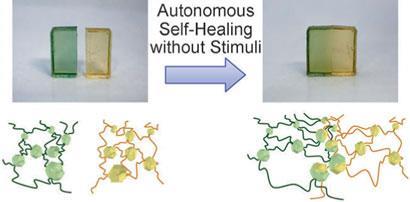
Hideyuki Otsuka’s team at of Kyushu University came up with a new solution using a cross-linking unit - diarylbibenzofuranone, DABBF - that can split to form a pair of radicals. ’Our system achieved "autonomous" self-healing and does not require any stimulation,’ he says.
The neat trick is that the DABBF units are in equilibrium, constantly breaking into arylbenzofuranone radicals and then reforming. This means that once damaged, the material just has to be put back in contact with itself to heal. As the DABBF units break and reform, the two surfaces will slowly bind back together. After 24 hours, the gel is as strong as it was before it was damaged.
Stephen Craig, who works on self-healing materials at Duke University, US, agrees that the underlying radical chemistry used here ’has some potential practical advantages’. But, he adds that ’the ultimate success of any system will increasingly be judged by the applications that it enables, and it will be interesting to see what the authors have in mind’.
Otsuka says he sees the system being used in fields such as ’space materials and artificial organs that have difficulty repairing’. At the moment the gel is not biocompatible because of the organic solvent used but Otsuka says he hopes that he can make the gel without these solvents in the future. First, he has to show that the DABBF units are not themselves toxic, particularly because radical species are often toxic.
’Preliminary results revealed that DABBF units do not show special toxicity,’ he says. ’We hope we can report on this topic after careful investigation in the near future.’
Laura Howes
References
K Imato et al, Angew. Chem., Int. Ed., 2011, DOI: 10.1002/anie.201104069






No comments yet