Molecular threads woven into flexible fabric
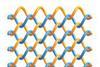
New covalent organic framework contains interlacing polymer chains that can slide past one another

New covalent organic framework contains interlacing polymer chains that can slide past one another