New carbodiphosphoranes break superbasicity record
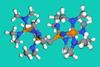
Chemists overcome synthetic challenges to make uncharged carbon molecules that sit at far end of THF basicity scale
Researchers have developed a new family of non-ionic carbon superbases featuring a carbodiphosphorane group. These air-stable structures are the most basic uncharged carbon bases to date