New silicon-based protecting group removable with blue light
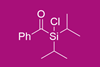
Benzoyldiisopropylchlorosilane protects primary, secondary and tertiary alcohols, and also works alongside other protecting groups
Researchers in Germany have created the first example of a silicon-based protecting group that is removed using visible light. The new protecting group will allow organic and materials chemists to protect alcohols under mild conditions and without needing UV light.
