Researchers in the US have identified an organocatalyst that can deconstruct a range of condensation polymers.1 The protic ionic salt catalyst is the first single catalyst to transform a mixture of consumer plastics into high-quality monomers.
Almost 79% of virgin plastics are estimated to enter landfills or gather elsewhere in our environment.2 ‘[A big challenge for recycling plastics] is that getting high quality, sorted post-consumer plastic is very labour and energy intensive,’ comments Julie Rorrer, an expert in chemically recycling waste plastics from the University of Washington, US, who was not involved in the research.
Now, Tomonori Saito and his research group from Oak Ridge National Laboratory, have developed a new strategy for tackling mixed plastic waste. It uses a protic ionic salt-based organocatalyst that selectively and sequentially deconstructs multiple condensation polymers while keeping other polymers, such as polyethylene and polypropylene, intact. ‘The study developed a technology that converts a conventionally unrecyclable mixture of plastic waste into useful chemicals, presenting a solution to combat the global plastic waste challenge,’ Saito tells Chemistry World.
The recent movement towards using organocatalysis, over metal-based catalysis, comes as these processes can be carried out under milder, more sustainable conditions. Organocatalysts are also more air and moisture stable and have lower toxicity, making them easier to handle.
The organocatalyst developed by Saito’s team comprises two components, trifluoracetic acid (TFA) and triazabicyclododecane (TBD). It deconstructs condensation polymers via a dual-activation mechanism with ethylene glycol acting as a nucleophile. The TFA component provides high conjugate basicity, which enhances the reactivity of the nucleophile in degrading the polymer. The protonated TBD component (TBDH+) coordinates to a carbonyl group in the polymer chain to increase the reactivity and weaken the bond that needs to be broken. Reactivity differences of functional groups within different polymer structures means that the organocatalyst can sequentially breakdown polymer mixtures. Gradually increasing the reaction temperature deconstructs each polymer at a different point in the process, making it easier to separate the resulting monomers.
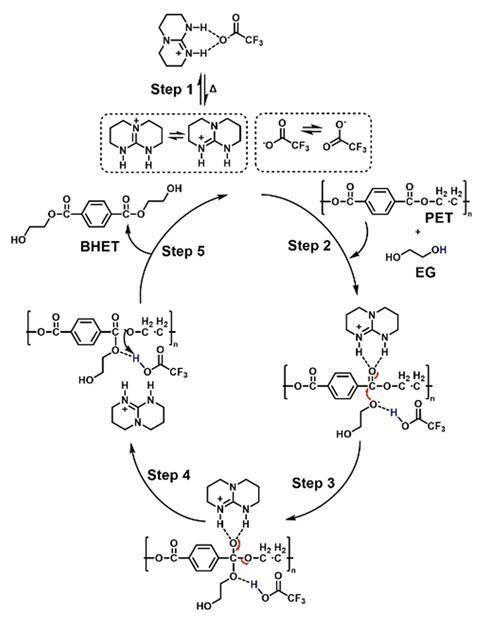
The team demonstrated the catalyst’s selectivity by applying it to mixtures of consumer plastics, polyethylene terephthalate bottles, polyethylene bags and mixed fabrics. It completely converted all condensation polymers into their respective monomers within two hours. Meanwhile, ‘other plastics such as polyolefin and cellulose are unreactive to this catalytic deconstruction process, which allows them to be kept intact and be readily separated from the mixture,’ adds Saito.
Saito’s team performed a life cycle assessment on their recycling strategy. It showed that synthesising polymers such as polyethylene terephthalate, polycarbonate, polyurethane and polyamide from the deconstructed monomers resulted in up to 95% less greenhouse gas emissions and required up to 94% less energy input than conventional methods.
As various companies already use similar solvolysis chemical recycling processes, the team hope that their process can be easily scaled up. However, Rorrer adds that when scaling up, ‘additional factors will come into play that you’ll have to build into those life cycle and economic analysis models to make sure that the process both pays for itself but is also good, or better, for the environment than the alternative in a linear plastic economy.’
References
1 M Arifuzzaman, Mater. Horiz., 2023, 10, 3360 (DOI: 10.1039/d3mh00801k)
2 R Geyer, JR Jambeck, KL Law, Sci. Adv., 2017, 3, e1700782DOI, DOI: 10.1126/sciadv.1700782













No comments yet