A new class of peptides that insert themselves into the cell membrane and form an open pore has been created.
A new class of peptides that insert themselves into the cell membrane and form an open pore has been created.
One of the most important functions of the biological membrane is to provide a protective barrier between a cell and its outside environment. Protein channels run across the membrane and maintain the vital salt concentration inside of the cell by opening and closing to regulate the transport of ions. A new class of antimicrobial peptides - the hydraphiles - exploits this circumstance.
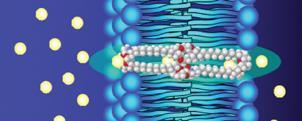
The peptides were designed by George Gokel and co-researchers at Washington University, US. Patch-clamping experiments on living cells showed that the compounds insert themselves into the cell membrane and form an open pore or channel. This results in the rapid and unregulated flow of ions across the membrane, producing a fatal osmotic stress.
Depending on the structure of the peptide, the hydraphiles can be made specifically toxic to bacterial, yeast or mammalian red blood cells.
Although the potency of the current range of compounds rivals those of conventional antibiotics, both synthetic and natural, ’flexible synthetic access to these compounds allows structural components.to be optimised to maximise toxicity.’
Michael J Spencelayh
References
W M Leevy, S T Gammon, T Levchenko, D D Daranciang, O Murillo, V Torchilin, D Piwnica-Worms, J E Huettner and G W Gokel, Org. Biomol. Chem., 2005, 3, 3544 (DOI: 10.1039/<MAN>b508157b</MAN>)





No comments yet