Stressed ligands switch catalysis selectivity
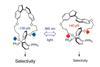
Switchable ligand flips between strained and relaxed forms altering reaction enantioselectivity

Switchable ligand flips between strained and relaxed forms altering reaction enantioselectivity