Weak gold-gold interactions in organic complexes affect the systems' emission spectra and could lead to a new type of sensor.
Researchers at Queen’s University Belfast are unravelling some of the fundamental chemistry surrounding a key but poorly understood aspect of the coordination chemistry of gold - the weak ’aurophilic’ interactions between adjacent atoms of Au(I) in organic complexes.
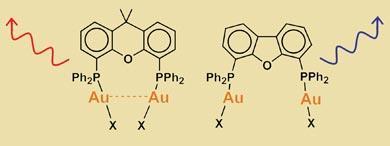
Cristina Lagunas and colleagues have demonstrated the relationship between aurophilic interactions and luminescence in a range of organic complexes. The work could lay the foundations for the development of gold-based organic sensors.
The Belfast chemists have been working with luminescent diphosphine compounds in which a phosphorus atom sits at either end of a hydrocarbon backbone, and a gold atom is attached to each phosphorus atom. By lengthening or shortening the backbone, the proximity of the gold atoms to one another can be controlled - allowing strong or weak aurophilic interactions to occur.
The team showed that when the molecule is too long to permit aurophilic interactions, the system emits blue light. As the backbone is shortened, aurophilic contact is made and the wavelength of the emitted light lengthens towards red.
In their most recent, unpublished, work the team has systematically bolted a variety of ligands on to the gold atoms to observe their effect on the emission spectrum. ’What we have seen is that if the gold atoms are separated by a distance of 3 ? or less, the aurophilic interaction is stable enough to withstand any interference by the ligand,’ Lagunas told Chemistry World. ’However, beyond this distance the interactions are weaker and more susceptible to interference by ligands, which alter the emission spectrum.’
This finding opens the possibility of creating sensors based on Au(I) complexes; the distance between the gold atoms could be tuned in such a way to make the aurophilic interaction - and thus emission spectrum - susceptible to changes by specific ligands. Simon Hadlington






No comments yet