Forcing fluorines into shape
Sometimes unnatural molecules can be more challenging to synthesise than natural metabolites
For some, the phrase ‘total synthesis’ conjures images of Robert Woodward lighting another cigarette as he works into the evening on his groundbreaking preparation of vitamin B12. For others, KC Nicolaou’s searing yellow structures of polycylic brevetoxins on infinitely black slides are a most vivid image, while for a younger generation, the term is inherently linked to Phil Baran’s eternal suntan and strategic masterclasses.
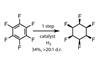
One unnatural target that had long been pursued, due to its expected physical properties, is all-cis 1,2,3,4,5,6-hexafluorocyclohexane – a facially polarised cyclohexane now known to be one of the most polar organic molecules in existence. The latest synthesis – in a single step from hexafluorobenzene – is likely to hold the title of ‘best’ for some time!
