Researchers have designed, built and programmed a chemical-handling robot that can screen and predict chemical reactivity using machine learning. Based on the autonomous system’s predictions, the team found four novel reactions, demonstrating its potential to discover reactions quickly.
Countless chemical reactions are known and many different pathways can lead to a desired molecule. To find the best pathways, discovering new chemical reactivity is crucial to make the processes that produce chemicals, pharmaceuticals and materials more sustainable, environmentally-friendly and efficient. However, discovering new reactions is usually an unpredictable and time-consuming process that’s constrained by a top-down approach involving expert knowledge to target a particular molecule.
Chemistry World got a sneak peak at the Cronin lab’s robot chemist when we visited Glasgow University in 2016
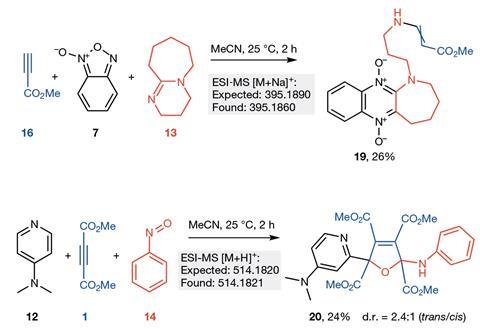
Now, Lee Cronin’s lab at the University of Glasgow, UK, has created an organic synthesis robotic AI system that can quickly explore the reactivity of a set of reagents from the bottom-up with no specific target. By just performing around 10% of 969 possible reactions from a set of 18 reagents the autonomous system was able to predict with 86% accuracy the reactivity of the remaining 90% of reactions. It then automatically performed more rounds of experiments based on the reactivity data it had gathered, continually updating the database, and in doing so discovered four new reactions. These were followed up manually by the team to isolate and characterise the new compounds.
‘I’m really surprised that the system was able to discover new reactions and molecules and especially the structure of one of the molecules is really strange and unexpected,’ says Cronin. ‘This is proof of principle that target-free organic discovery and synthesis can yield really unexpected and perhaps even very novel results that could fundamentally change how we go about looking for new reactions.’
Robot scientist
The robot can perform up to 36 experiments per day – around 10 times more than a human. It builds a database of reaction information by first randomly selecting and combining different reagents from a given set. Samples of each mixture are subsequently analysed for reactivity by obtaining their spectra in real-time using built-in sensors, including NMR, infrared and mass spectrometry.
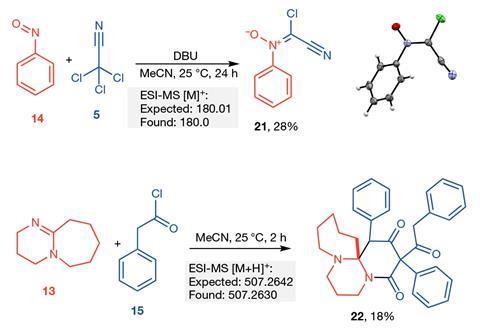
A machine learning algorithm designed to recognise reactivity based on differences in spectra from the original starting reagents then classifies reaction mixtures as either reactive or unreactive. This data is then fed back to the robot to decide the next round of experiments. This means the system is able to build-up a picture of how the reagents react with each other and determine if a reaction will be successful or not. Consequently, the system can prioritise the reactions it thinks have the greatest potential. Using this approach, the robotic chemist discovered four new reactions – two two-component reactions and two three-component reactions.
You might also enjoy:
Art or automation? How robotics is set to transform chemistry
Since the system has no prior information – except the spectra of the starting reagents needed by the algorithm – it builds a database from scratch. ‘This is desirable for reaction discovery since it removes bias and allows the system to more freely explore,’ explains Cronin. ‘It was tremendously exciting to see the system autonomously navigate and “explore” for the first time since chemical automation systems normally only do what they are told and are not “creative”,’ he says.
Disruptive chemistry
‘One of the remarkable aspects of this study is the integration of automated chemical synthesis, multiple modes of automated product analysis and machine learning into a closed loop that autonomously seeks a complex chemical objective,’ comments Marty Burke, who investigates small molecule synthesis at the University of Illinois at Urbana–Champaign, US. ‘If such closed loops can, in the future, help solve some of the most challenging but potentially transformative problems in chemical reactivity they will have a disruptive impact on the process and accessibility of molecule making.’
Pablo Carbonell, who uses machine learning for synthetic biology at the University of Manchester, UK, is impressed too. ‘Artificial intelligence-based synthesis robots will certainly lead the automated design and optimal screening of next generation organic synthesis,’ he says. ‘Future developments could combine the use of machine learning to efficiently explore the reaction space, as presented in this work, with deep learning algorithms that extract chemical information in order to predict novel organic synthetic routes.’
Cronin is now planning to increase the scope of the system’s reaction conditions and enable it to perform multi-step processes. ‘We wish to discover some entirely new classes of reaction that challenge our current structure and bonding theory,’ he says. ‘We are also looking to add in assays that connect the search to a desirable discovery such as a drug target or material property.’
References
J M Granda et al, Nature, 2018, DOI: 10.1038/s41586-018-0307-8










No comments yet