The first solution synthesis of the elusive ‘Clar’s goblet’ structure – first proposed over 50 years ago – has provided the perfect platform to prove spin entanglement between non-bonded electrons. Studying spin pairing at this scale could increase our understanding of quantum chemistry, as well as accelerate applications in quantum computing.
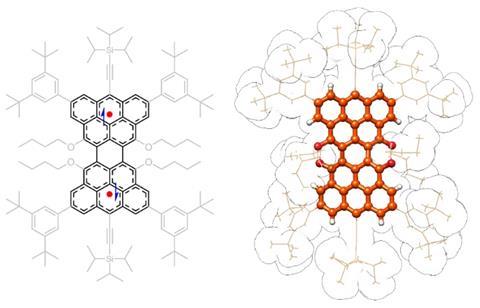
‘Clar’s goblet is a bowtie-shaped [structure] first proposed by Erich Clar in 1972,’ explains lead author Jishan Wu, from the National University of Singapore. ‘It’s characterised by the presence of two unpaired electrons, each … localised in one half of the molecule.’ However, it’s precisely the unpaired electrons that make the molecule extremely unstable, and until now its synthesis in solution seemed impossible.
‘Fasel, Feng and co-workers managed to generate single molecules of the Clar’s goblet on surface, under ultra-high vacuum conditions [a few years ago],’ explains Diego Peña, an expert in the synthesis of nanographenes at CIQUS in Santiago de Compostela, Spain. He says this new approach ‘is truly impressive’.
The study suggests that, although spatially separated by a distance of 8.7Å, ‘the two unpaired electrons in Clar’s goblet … exhibit antiferromagnetic coupling’, explains Wu. This is fundamental for molecular quantum technologies, which rely on connecting and coupling spin centres with interactions like coherence and entanglement. ‘Clar’s goblet demonstrates that nanographene radicals [could] serve as a molecular spin system.’
This effect is a consequence of the ‘peculiar shape and carbon connectivity’ of this ‘iconic bowtie-shaped polycyclic aromatic hydrocarbon’, says Peña. Although this ‘was previously predicted by theory and demonstrated in single molecules [with] on-surface experiments, this work now proves this unusual behaviour in solid and solution phases’. The researchers included an ‘impressive characterisation, including x-ray crystallographic analysis, electron paramagnetic resonance, DFT calculations … and spin dynamic studies’, he adds. Overall, the study shows ‘the two electrons can feel the existence of each other despite the distance’, which is a unique and unconventional magnetic effect in molecules like nanographenes.
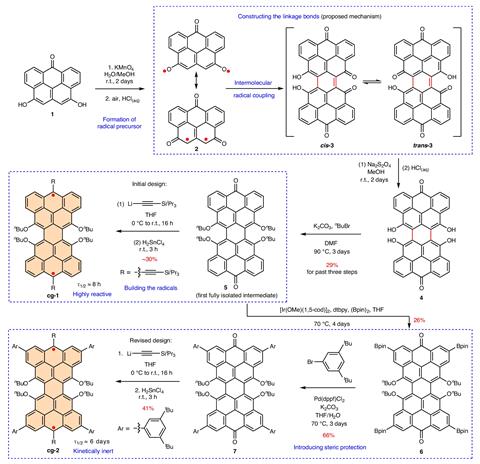
The synthesis in solution was almost a serendipitous discovery, according to Wu. ‘We discovered that the dioxyl-substituted diradical precursor could undergo [an intermolecular] radical coupling to construct the conjugated backbone of Clar’s goblet,’ he says. ‘This … marked the beginning of our journey.’ After fusing the fragments of the bowtie, the team introduced several stabilising substituents – the steric interactions boost inertness, making the molecule easier to isolate. ‘We believe the ingenious design of the synthetic strategy was key,’ adds Wu.
‘More than half a century after Clar’s original proposal … this study delves into our knowledge on spin entanglement in carbon-based materials and facilitates the use of nanographenes for quantum devices,’ explains Peña. The authors also managed to make ‘milligram quantities of such an exotic molecule, which could facilitate its integration into quantum devices’.
The spelling of Erich Clar’s name was corrected on 9 April 2025
References
T Jiao et al, Nat. Chem., 2025, DOI: 10.1038/s41557-025-01776-1















No comments yet