A new compound that binds to a part of the µ-opioid receptor (µOR) could be a useful tool in preventing deaths from opioid overdose. The molecule works in conjunction with naloxone, boosting its effectiveness seven-fold.
Naloxone is an effective treatment for opioid overdose. However, larger and repeated doses are needed in response to overdoses from more potent synthetic opioids such as fentanyl.
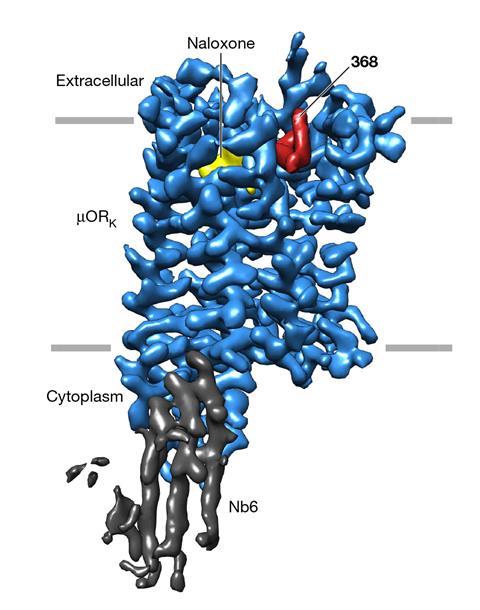
In a search for a potent alternative to naloxone, researchers screened a large DNA-encoded chemical library and identified a potential candidate that was highly selective for the µ-opioid receptor. This new compound – codename 368 – is a negative allosteric modulator that binds to the µOR but at a different site to opioids. The researchers explained that, until now, selective potent negative allosteric modulators for the µOR had remained elusive.
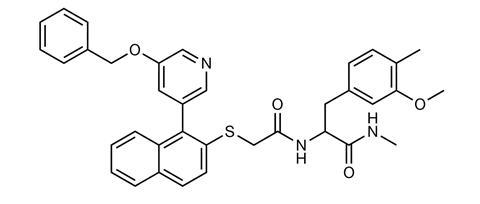
The researchers found that when 368 was bound to the µOR, it enhanced the binding affinity of naloxone, boosting its potency by over seven-fold. Observations made using cryo-electron microscopy showed that 368 worked cooperatively with naloxone to potently block opioid agonist signalling.
In vivo mouse models revealed that the addition of 368 meant that lower doses of naloxone could be used to effectively inhibit the effects of morphine and fentanyl, while minimising withdrawal symptoms.
Further screening of other negative allosteric modulators could help uncover their mechanism of action and improve the effectiveness of these compounds, the researchers write.
References
ES O’Brien et al, Nature, 2024, DOI: 10.1038/s41586-024-07587-7















No comments yet